Science of mitochondrial donation
3.1
Chapters one and two of this report briefly covered what mitochondria
are and how mutations in mitochondrial DNA[1]
(mtDNA) can cause mitochondrial disease.
3.2
Mitochondrial donation techniques allow the mother's mutated mtDNA, which
will lead to the potential formation of a mitochondrial disease, to be
substituted for a donor's healthy mtDNA.
3.3
This chapter will cover the mitochondrial donation techniques that could
be used to prevent the transmission of this mutated mtDNA to the children of
women living with a mitochondrial disease and will consider some of the
scientific risks associated with these techniques.
Mitochondrial donation techniques
3.4
Throughout the course of this inquiry, the committee was advised there were
four possible methods of mitochondrial donation: maternal spindle transfer,
pronuclear transfer, polar body transfer and germinal vesicle transfer. These techniques
are outlined below.
Maternal spindle transfer
3.5
Maternal spindle transfer is a technique in which the spindle shaped
group of chromosomes containing the mother's nuclear DNA, known as the
'maternal spindle', is extracted from one of the mother's eggs (oocytes) and
transferred to an unfertilised donor egg from which the maternal spindle has been
removed and that contains healthy mtDNA.[2]
3.6
Once the maternal spindle has been transferred to the donated egg with
the healthy mtDNA, the egg is fertilised with the father's sperm and then
implanted into the uterus in a manner similar to other in vitro fertilisation
(IVF) techniques.[3]
3.7
By removing the maternal spindle and inserting it into an egg with
healthy mtDNA, the resulting offspring will receive the 22 000 base pairs
of nuclear DNA from the parents, but will have the 37 base pairs of healthy mtDNA
from the oocyte donor.[4]
3.8
A visual diagram of a maternal spindle transfer is included below.
Figure 3.1—Maternal spindle
transfer
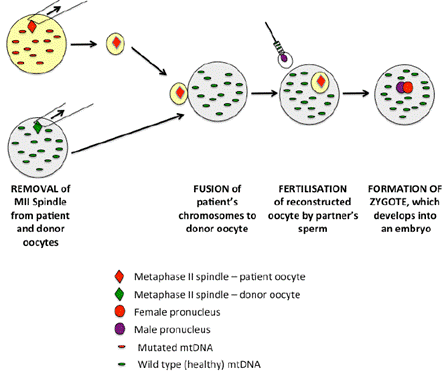
Source: Professor Justin St John, Submission 31,
[p. 7].
Current status
3.9
In the United Kingdom (UK), maternal spindle transfer is one of two
methods that have been legalised by The Human Fertilisation and Embryology
(Mitochondrial Donation) Regulations 2015 (UK).[5]
3.10
As it is not the method that is preferred by the clinic that is
currently licensed to conduct mitochondrial donation, less research has been
conducted using this method. Professors David Thorburn, John Christodoulou,
Carolyn Sue, John Carroll, Mike Ryan and Aleksandra Filipovska advised the
committee that this technique has been used successfully in Macque monkeys by a
research group in Oregon in the United States of America (USA) and has led to
one live birth.[6]
However, at this stage, maternal spindle transfer has not yet 'been fully
optimised for human eggs'.[7]
3.11
In Australia, the National Health and Medical Research Council (NHMRC)
has advised the committee that research on maternal spindle transfer is
currently prohibited by section 13 of the Prohibition of Human Cloning for
Reproduction Act 2002 (Cloning Act) which provides:
A person commits an offence if:
- the person intentionally creates or develops a human
embryo by a process of the fertilisation of a human egg by a human sperm
outside the body of a woman; and
- the human embryo contains genetic material provided by
more than 2 persons.
Penalty: Imprisonment for 15 years.
3.12
The maternal spindle transfer method is prohibited because the process
requires fertilising a human egg, creating an embryo with genetic material from
more than two persons.
Pronuclear transfer
3.13
The same difficulty is not experienced with the pronuclear transfer
technique because the egg is fertilised, prior to the nuclear DNA transfer
occurring.[8]
When an egg is fertilised and becomes a zygote, two pronuclei are formed (one
from the mother and one from the father) containing the parents' nuclear DNA. For
pronuclear transfer, a second zygote must be created from a donor egg and the
father's sperm. The two pronuclei from the first zygote are removed and
transferred to the donor zygote with healthy mtDNA.[9]
The donor zygote, which needs to be at the same stage of development, has had
its pronuclei removed to facilitate the transfer.[10]
3.14
A visual diagram of pronuclear transfer is included below.
Figure 3.2—Pronuclear transfer
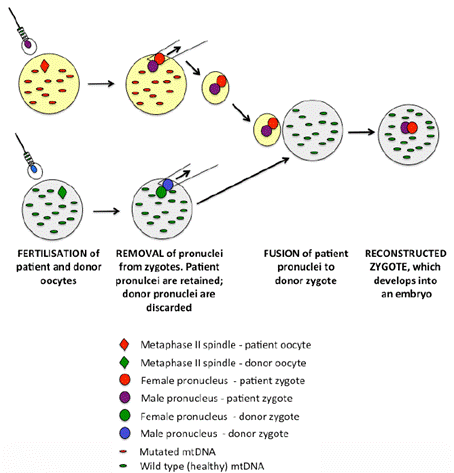
Source: Professor Justin St John, Submission 31,
[p. 7].
Current status
3.15
Pronuclear transfer is the second method that has been legalised in the UK.[11]
3.16
Pronuclear transfer is the technique that has been investigated in
greater depth by the clinic at the University of Newcastle-upon-Thyne in the UK
which holds the licence from the Human Fertilisation Embryology Authority
(HFEA) to perform mitochondrial donation.[12]
3.17
In Australia, the NHMRC has advised the committee that a licence to
research the pronuclear transfer technique can be granted under current
legislation because the egg has already been fertilised prior to its transfer
to the donor egg.
3.18
It is, however, subject to two other restrictions: first, the Cloning
Act restricts the development of any embryo outside the body of a woman to a
period of 14 days.[13]
The NHMRC explained that it was possible for some research to be conducted:
The creation of the reconstructed embryo could also be licensed
under paragraph 20(1)(c) and it could be maintained in culture to assess the
success of the procedure provided it was discarded before 14 days had elapsed.[14]
3.19
The second restriction precludes pronuclear transfer, or any other form
of mitochondrial donation technique, from being used for reproduction. This
restriction arises because any embryo containing the genetic material of more
than two persons is considered to be a prohibited embryo for the purposes of
the Cloning Act and cannot be implanted into a uterus for development into a
foetus.[15]
Polar body transfer
3.20
A third possible method for mitochondrial donation is known as polar
body transfer. There are two different techniques for polar body transfer.
3.21
During each menstrual cycle, some eggs are 'selected' for maturation and
growth. As part of this process, the cell divides and leads to the formation of
a secondary egg that contains mostly nuclear DNA and very little cytoplasm,
which is the surrounding material in which the mitochondria are found. This is
known as the first polar body.
3.22
The first polar body transfer technique extracts the first polar body,
which sits outside of the main egg, and fuses it to an unfertilised egg that
has had its maternal spindle removed. The reconstituted egg is then fertilised
by the patient's partner's sperm.
3.23
A visual representation of the first polar body technique in comparison to
the maternal spindle transfer technique is included below.
Figure 3.3—First polar body
transfer
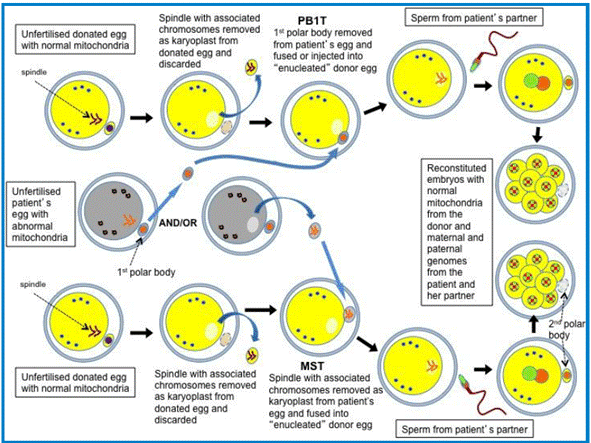
Source: HFEA, Review of the safety and efficacy of polar
body transfer to avoid mitochondrial disease, October 2014, additional
information received 30 May 2018, p. 17.
3.24
The second polar body is formed during fertilisation when the egg splits
again. The second polar body transfer technique involves extracting the second
polar body after fertilisation and transferring it to a newly fertilised egg
that has had its maternal nuclear DNA removed.[16]
The second polar body is then fused into the reconstituted egg.
3.25
A visual representation of second polar body transfer and how it
compares to pronuclear transfer is included below.
Figure 3.4—Second polar body
transfer
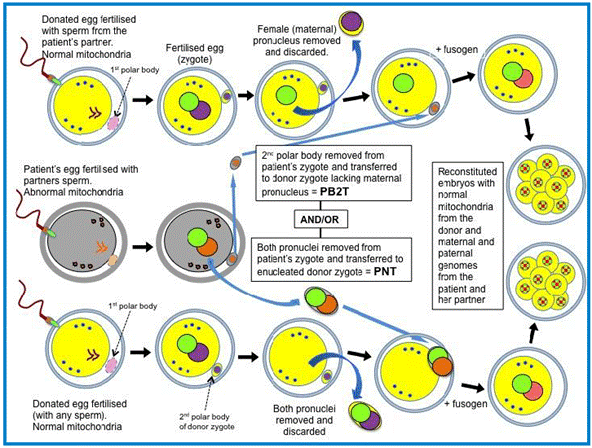
Source: HFEA, Review of the safety and efficacy of polar
body transfer to avoid mitochondrial disease, October 2014, additional
information received 30 May 2018, p. 18.
3.26
There may be advantages to using polar body transfer over maternal
spindle transfer or pronuclear transfer because it may:
-
reduce mtDNA carryover;
-
reduce the risk of leaving chromosomes behind in maternal spindle
transfer; and
-
be possible to carry out both polar body transfer and either
maternal spindle transfer or pronuclear transfer.[17]
3.27
However, at this stage it does not appear that polar body transfer
techniques have been as advanced as some of the other methods.
Current status
3.28
In the UK, polar body transfer cannot legally be used in clinical practice.
A safety and efficacy review of polar body transfer conducted by the HFEA found
that while polar body transfer techniques were developing quickly, they were
still at an early stage.[18]
3.29
Professors Thorburn, Christodoulou, Sue, Carroll, Ryan and Filipovska
advised the committee that they understand that the technique is still at the
preclinical study stage and further work is still required to understand and
optimise the procedure.[19]
3.30
The committee is not aware of polar body transfer research being conducted
in Australia.
Germinal vesicle transfer
3.31
Another possible technique pioneered by Professor Justin St John is
called germinal vesicle transfer. This method, which was not well-known by many
of the submitters, is similar to maternal spindle transfer except that it uses
an egg that is at an earlier stage of development.
3.32
In germinal vesicle transfer, the germinal vesicle (which will develop
into the maternal spindle) is extracted from an egg that is at an earlier stage
of development and the germinal vesicle is allowed to develop in vitro.[20]
3.33
Professor St John explained that there may be benefits to using this technique
because it would not require the woman to undergo superovulation protocols and may
give the chromosomes 'a bit longer to readjust to the new environment they are
in'.[21]
3.34
Currently, there is little data of germinal vesicle transfer. [22]
3.35
Professors Thorburn, Christodoulou, Sue, Carroll, Ryan and Filipovska
advised the committee that success rates using this technique are currently low
that 'the need to retain the egg's supporting cells will create technical
challenges'.[23]
3.36
A visual diagram of the germinal vesicle transfer technique is included
below.
Figure 3.5—Germinal vesicle
transfer
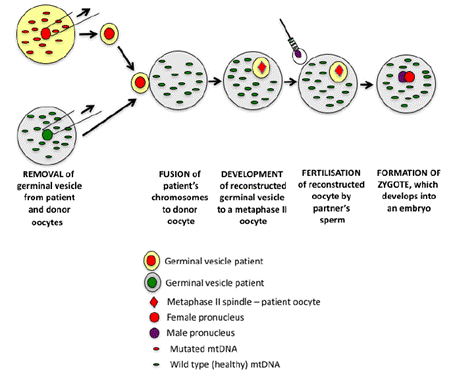
Source: Professor St John, Submission 31, [p. 9].
Current status
3.37
Germinal vesicle transfer has not been legalised in the UK for clinical
implementation. In Australia, the same restrictions are likely to apply as
currently apply to maternal spindle transfer.
Potential risks
3.38
Any emerging reproductive technology includes a degree of risk.[24]
Witnesses and submitters to the inquiry explained the risks that may exist with
mitochondrial donation techniques.
3.39
The threshold question for the committee's consideration of the matter
was whether mitochondrial donation techniques are considered to be safe to
perform on human embryos that will develop into live babies.
3.40
Submitters to the inquiry were largely of the opinion that mitochondrial
donation is now safe to perform.[25]
3.41
Before mitochondrial donation was legalised in the UK, these techniques
were subject to four scientific reviews. The Wellcome Trust, a UK based
charitable foundation that funds mitochondrial disease research, told the
committee that scientific reviews conducted prior to legalisation concluded
that the techniques were safe:
Safety of the techniques is, and will always be, of paramount
importance and has received unprecedented scrutiny. On three separate occasions
the HFEA's specially convened independent Expert Scientific Review panel
examined the safety and efficacy of mitochondrial donation. The panel reported
that they found no evidence to suggest that the techniques are unsafe for
clinical use, and concluded that both techniques have the potential to be used
in patients with mitochondrial disease.[26]
Box 3.1—UK
Scientific Reviews
Before mitochondrial donation
was legalised in the UK three scientific reviews were undertaken by an expert
panel convened by the regulator, the HFEA, to assess the safety and efficacy of
the techniques.
The third scientific review was
completed in 2014. The 2014 review recommended that:
- additional experiments
needed to be conducted to corroborate and improve the efficiency of the
maternal spindle transfer technique;
- additional experiments needed
to be conducted to compare pronuclear transfer ooyctes with intracytoplasmic
sperm injection oocytes; and
- consideration should be
given to mtDNA haplogroup matching.[27]
The fourth scientific review
was completed in 2016. The 2016 review was conducted to update the 2014
scientific review and to consider whether the recommendations made in that
report had been met. The review considered that good progress had been made on
each recommendation. In addition, it recommended that clinicians carefully
select patients, conduct prenatal testing and follow up and maintained the
recommendation to use haplogroup matching as a precautionary step.[28]
3.42
A similar conclusion has been reached by Australian experts. Professor John
Carroll, Director of the Monash Biomedicine Discovery Institute at Monash
University told the committee that the evidence did not indicate that there
were any serious safety concerns:
...a good deal of research has been done, and to date there's
really very little evidence for serious safety concerns and certainly nothing
that comes anywhere near close to the impact that genetic disease has. Being
able to assess the risks associated with the procedure with the alternative
outcome, I think there's very little doubt in my mind, at least, that they're
very well balanced, and we should be able to proceed with investigating the
treatment.[29]
3.43
Professor John Christodoulou, Chair of Genomic Medicine in the
Department of Paediatrics at the University of Melbourne told the committee
that he was unaware of any evidence that pointed to there being significant
risks to a child born of a mitochondrial donation:
There has been some theorizing that mitochondrial donation
through proposed epigenetic mechanisms, or as a consequence of not using mtDNA
haplogroup matched donor egg cells for the procedure, could lead to untoward
effects on the health of the embryo or the child after birth. However, I am
aware of no such evidence supporting the notion that there would be any significant
risks to children born following mitochondrial donation.[30]
3.44
Some submitters though were more cautious about declaring the techniques
as being safe to use. The submission from the NHMRC noted that although one
child is known to have been born in Mexico using the maternal spindle transfer
technique, his mutation load is currently unknown because:
...his parents have requested no more testing for mtDNA unless
there is a clinical benefit. Consequently, it may be difficult to assess the
long-term success of the procedure.[31]
3.45
Professor St John considered that additional studies were required on
large animal models to test the consequences of mtDNA carryover and test the
effects of using eggs with different haplotypes.[32]
These issues are considered in turn below.
Carryover of mutated mtDNA
3.46
Some submitters and witnesses to the inquiry expressed concerns about
the potential effect of carrying over mutated mtDNA to the reconstituted donor
egg during the transfer process.[33]
3.47
Professor Christodoulou explained that when the nucleus of the cell is
transferred from one egg to another, a certain amount of the mutated mtDNA may
be carried over:
The process involves removing a nucleus and then implanting
that nucleus into the egg cell that's had the nucleus removed and has the
mitochondria. In the early days, the process of removing the nucleus, as part
of it, took a number, or a proportion, of mitochondria—and therefore mitochondrial
DNA—along with it in that process. That's that sort of carryover phenomenon.[34]
3.48
Some submitters expressed concern that if mutated mtDNA is transferred
to the donor egg, the child may still end up with a mitochondrial disease.
3.49
According to Professor Christodoulou, the expert committee for the HFEA
recommended that mtDNA carryover rates should not exceed two per cent and
should be no greater than 10 per cent per embryo.[35]
A number of witnesses who provided evidence to the committee, including
Professor Christodoulou, endorsed the two per cent figure as representing a
safe level below which a child was unlikely to develop a mitochondrial disease.[36]
3.50
This figure is considered to be a safe level because, as Associate
Professor Damian Dowling from the School of Biological Sciences at Monash
University explained in his submission, mutations in mtDNA do not generally
cause mitochondrial disease until the mutated mtDNA comprises 70–80 per cent of
the pool of mtDNA.[37]
However, Associate Professor Dowling suggested that even a small amount of carryover
may present a risk to the child. The risk may exist because mtDNA cell numbers
are not static across a person's life and experimental studies have shown that
the unhealthy mtDNA cells can 'outcompete' the healthy mtDNA cells:
Experimental studies in flies, yeast, worms, and human cell
lines have shown that defective mtDNA molecules often proliferate more rapidly
than healthy molecules, and can thus, somewhat ironically, outcompete their
healthy mtDNA counterparts...This means that mitochondrial disease could plausibly
reemerge in children born to the technique, or in the children of daughters
born to this technique. [38]
3.51
Professor St John and Dr Ian Trounce both noted that there have been
some studies in which the original mtDNA outcompeted the donor's mtDNA to
become the dominant mtDNA in the population.[39]
Murdoch Children's Research Institute and Victorian Clinical Genetic Services
and Professor Mary Herbert also noted that in
15‒20
per cent of cases, stem cells tested after mitochondrial donation showed
that the mtDNA had reverted to the maternal mtDNA.[40]
3.52
Professors Thorburn, Christodoulou, Sue, Carroll, Ryan and Filipovska
doubted whether reversion to the maternal mtDNA would be seen in live babies,
noting significant differences between the long-term culture of stem cells and
live births:
...embryonic stems cells are considered a poor proxy for normal
development in the womb...The state of pluripotency, which allows stem cells to
proliferate indefinitely in cell culture, lasts for only a few days during
normal development.[41]
3.53
Furthermore, international evidence suggests that there is not
significant drift overtime and the amount of mutated mtDNA present at the
eight-cell stage and the prenatal diagnosis stage is consistent with the 'level
found in multiple tissues at birth'.[42]
3.54
Many submitters were of the opinion that mtDNA carryover could be
managed and minimised. In its submission, the Wellcome Centre for Mitochondrial
Research, the clinic that currently holds the licence to conduct trials from
the UK's HFEA, told the committee that in its initial studies the level of
carryover was minimal:
The study revealed that human PNT embryos had the potential
for onward development and importantly, that the level of mtDNA co-transferred
with the nuclear DNA during the procedure was minimal (<2% on average). This
is well below the level of mutant mtDNA associated with clinical symptoms and
led us to conclude that PNT had the potential to prevent transmission of
mitochondrial disease.[43]
3.55
Since then, additional studies have been done to examine whether there
was the potential for the mutated mtDNA to increase to substantial levels. In a
joint submission, the Murdoch Children's Research Institute and Victorian
Clinical Genetic Services advised the committee that additional research had
been conducted in the UK which found that the levels of mtDNA did not increase
provided the original transfer was kept to below two per cent of the mutated
mtDNA:
Additional safety experiments were performed to determine
whether there was any potential for mitochondrial DNA carry-over to result in
the original mitochondrial DNA from the mother's egg increasing back up to
substantial levels. Their data showed that this did not happen so long as the
procedure ensured no more than 2% of the mother's mitochondrial DNA was present
in the embryo after mitochondrial transfer.[44]
3.56
Professor Carroll advised the committee that he considered that, if good
techniques are used, the risks of the baby acquiring a genetic disease were
low:
My view is that, once you're doing the mitochondrial
procedure, the risks of carryover that are sufficient to contribute to the baby
having any genetic disease is so low that I don't think it's a necessary part
of the procedure... I think that the mitochondrial donation procedure leads to
so few mitochondria, using good techniques, being donated to the new embryo
that it's unlikely to be propagated.[45]
3.57
The Fertility Society of Australia (Fertility Society) also acknowledged
that, whilst a small risk exists, it is worth taking if it means that a child
will not be born with a fatal disease:
Based on the scientific advice of our membership, we believe
that the balance of safety versus risk has been addressed. There is no question
that new technology does occasionally bring negative results, but given the
fatal nature of Mitochondrial disease we believe that miniscule risk is worth
taking.[46]
The potential of sex-selection
3.58
As noted above, some researchers have raised a risk of mitochondrial
disease re-emerging in the children or the children of daughters born to
mitochondrial donation techniques because of mtDNA carryover.[47]
3.59
Dr Peter McCullagh, a British medical practitioner who followed the
developments in the UK, noted that there had been a proposal to restrict
clinics to selecting males embryos for clinical implantation to mitigate this
risk:
To ensure that the mutated mtDNA is not transmitted to any
children leading to a risk of transgenerational impacts, it has been proposed
that licences to undertake mitochondrial transplantation should be restricted
to British clinics which commit to gender selection for males. There have been
warnings that, even if the first generation of females is not clinically
affected, mitochondrial coded disease may nevertheless emerge in later ones.
Birth of a clinically normal infant may not necessarily guarantee similar
normality in the following generation.[48]
3.60
That argument found favour with the National Academies of Sciences,
Engineering and Medicine (NASEM) in the United States of America. A report by
the NASEM for the USA Food and Drug Administration recommended that initially
only male embryos should be transferred because there was a 'need to proceed
slowly and to prevent potential adverse and uncertain consequences of MRT [mitochondrial
replacement techniques] from being passed on to future generations'.[49]
3.61
Under the American proposal, female children would only be able to be
born after adequate follow-up and satisfactory findings in male children.[50]
This is because mtDNA cannot be inherited through the male line.
3.62
Mr Sean Murray from the Australian Mitochondrial Disease Foundation
(AMDF) told the committee that only selecting male embryos could be considered in
Australia as an interim safeguard measure:
I think the recommendation was made as a risk mitigation
there, because, as I explained before, in my situation I can't pass on my
mitochondrial DNA to my children, and I think that that's the rationale behind
that. So that could definitely be viewed as a safeguard measure while we figure
this out in more detail.[51]
3.63
Some submitters noted that a prohibition on implanting female embryos
would halve the efficiency of the techniques and would potentially require
women to undergo additional ovarian hyperstimulation to produce additional
eggs.[52]
3.64
Many of the scientists the committee spoke to considered that it was not
a necessary prohibition. Professor Thorburn noted that, even though it was
considered, the UK ultimately decided not to impose such a prohibition because
it was not considered to be necessary.[53]
3.65
Professors Thorburn, Christodoulou, Sue, Carroll, Ryan and Filipovska
advised the committee that, in their opinion, the same risks existed for male
and female embryos, meaning that there was no clear reason to prohibit the
implantation of female embryos.[54]
3.66
Some submitters considered that while a degree of risk exists, the question
whether to implant female embryos should be considered by the prospective
parents after counselling.[55]
mtDNA matching
3.67
As noted in chapter one, mtDNA is maternally inherited. Different people
have different mtDNA if they come from a different haplogroup (also sometimes
called a haplotype). A haplogroup corresponds to the common maternal origins of
the species. In humans, there are about 25 different major variations of the
mtDNA sequence and they largely correspond to continental population groups.[56]
3.68
A visual representation of the distribution of those haplogroups is
included below.
Figure 3.6—Haplogroup distribution
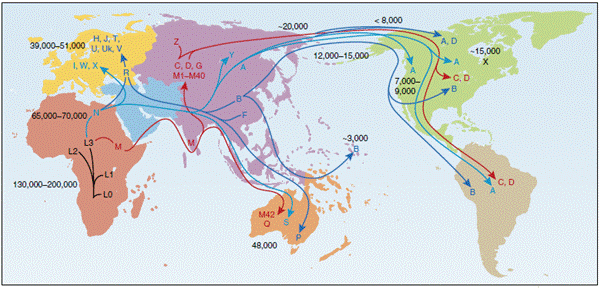
Source: Professor Robin Lovell-Badge, Submission 58—Attachment
1, p. 1061.
3.69
Submitters to the inquiry noted that a person's haplogroup can influence
a number of factors related to a person's health, including 'sperm motility,
infection resistance, susceptibility to neurodegenerative disease and ageing'.[57]
3.70
Some submitters have expressed concern that a failure to match the haplogroup
of the egg donor to the haplogroup of the mother's nuclear chromosomes may lead
to potential negative health effects on the child born of the technique. Professor
St John explained to the committee the nature of his concern:
We know from certain studies in both human and mouse models
that, if you increase the genetic distance between the source of the eggs from
which the chromosomes are coming and the donor egg itself, that can influence
the outcome and the phenotype of the offspring and the cells you're trying to
make.[58]
Incompatibility between different
haplogroups
3.71
There were two areas of concern where it was suggested there could be
negative consequences of unmatched haplogroups. The first of these was that
mtDNA from a different haplogroup may be inconsistent or incompatible with the
mother's chromosomes and this may in turn affect potential gene expression.
3.72
The first mechanism was raised by Professor St John, Associate Professor
Damian Dowling and Dr Ian Trounce who drew the committee's attention to studies
that primarily used mice and fruit flies and indicated that a failure to match
the mother's mtDNA haplogroup with the egg donor's mtDNA haplotype had the
potential to lead to changes in gene expression.[59]
3.73
Professor St John advised the committee that studies on mouse stem cells
had demonstrated using mtDNA from distantly related haplogroups could have an
effect on the health of the mouse.[60]
3.74
Associate Professor Dowling similarly observed that different variations
may alter the efficiency of the gene expression and 'in theory could therefore
affect individual performance'.[61]
He noted though that some mtDNA mutations could be beneficial in some
environments, for example the same mutation that causes the debilitating
Leber's Hereditary Optic Neuropathy (LHON) is the same mutation that can help
humans to survive at high altitudes in oxygen deficient environments.[62]
3.75
Other submitters were less certain that mtDNA matching was necessary. Professor
David Thorburn, Head of Mitochondrial Research and Diagnostic Laboratories at the
Murdoch Children's Research Institute and Victorian Clinical Genetic Services
questioned that the studies on mice and fruit fly would necessarily translate
to humans:
There are experiments in animals, particularly in inbred
mice, flies and worms, which suggest that there could be some degree of
incompatibility between distantly related haplogroups...They tend to be more
distantly related than humans are, and they tend to be inbred rather than outbred,
so you can measure outcomes very accurately. My personal view is that this is
unlikely to be an issue...[63]
3.76
Professor Thorburn also pointed to studies conducted with Macque monkeys
and the limited human evidence that is available to indicate that unmatched
mtDNA haplogroups have not led to health problems:
It's very reassuring that when this has been done in the
macaque monkeys, there hasn't been any evidence seen for this drift of maternal
versus donor haplogroups happening. Those monkeys have been healthy when
studied—at least the males have been shown to be fertile; the female has just
reached reproductive age I think—and the limited information of the one child
born from this technique in Mexico, which was a terrible regulatory process,
has had about the same amount of the mutation in cord blood and cheek wash and
all those sorts of non-invasive tests of tissues and placenta. So the available
evidence suggests that it hasn't been seen in primate models—monkeys or humans.[64]
3.77
Professors Thorburn, Christodoulou, Sue, Carroll, Ryan and Filipovska
note that while there is still some uncertainty, they consider that the 'likely
risks are relatively low'.[65]
Nuclear-mitochondrial interaction
3.78
The second area of concern where there may be negative consequences of
not matching haplogroups, is that there may be an evolutionary link or
interaction between the mtDNA and the nuclear DNA and breaking the link between
the two may lead to potential negative consequences.
3.79
Associate Professor Dowling explained in his submission that
evolutionary theory indicates that the mtDNA and the nuclear DNA have evolved
together. He suggested that in this way the nuclear DNA and the mtDNA were like
pieces of a jigsaw that would not necessarily be compatible with other mtDNA
and may cause negative health consequences in the offspring.[66]
3.80
Associate Professor Dowling expressed concern about this and pointed to
a number of studies that indicated there may be effects on humans as a result
of mitochondrial donation. However, he noted that it was uncertain whether
creating novel mitochondrial and gene combinations would lead to health benefits
or detriments:
The evidence to date suggests that it's more likely there
will be effects than no effects by creating novel combinations of mitochondrial
and nuclear genotype as mitochondrial donation will do. However, it's not
clear. We have, at this stage, no way to predict whether or not the effects
will actually be advantageous to the child and improve the performance of what
the child would have been in the event that it hadn't originally carried the
pathogenic mtDNA mutation, or whether it will result in a decrease in
performance in the child. The majority of evidence suggests the negative
effects are more common than the positive effects, but it can go either way.[67]
3.81
In particular, Associate Professor Dowling pointed to a 2018
meta-analysis study that suggested that 'humans showed stronger effects' than
other animals to mitochondrial donation.[68]
The meta-analysis referred to by Associate Professor Dowling estimated
'negative effects in at least one in every 130 resulting offspring born to the
therapy'.[69]
Professors Thorburn, Christodoulou, Sue, Carroll, Ryan and Filipovska submitted
that if that estimate is correct, the resulting risks are 'lower that the
approximately 3% risk for any couple of having a child with some kind of
genetic anomaly'.[70]
3.82
Other submitters disagreed that this would be a problem. The Progress
Educational Trust dismissed the suggestion that disrupting co-evolution could
lead to adverse consequences:
Some have argued that mitochondrial donation could disrupt
relationships that have developed between mitochondrial and nuclear DNA via
co-evolution, and that this could have adverse consequences. There is little
evidence for this view.
There have been experiments on animals where co-evolved
relationships between mitochondrial and nuclear DNA were deliberately
disrupted. However, this has only been shown to have a mildly adverse effect in
two situations, and neither of these situations is applicable to mitochondrial donation
in humans.[71]
3.83
The Progress Educational Trust was clear that while the nuclear genes
have an effect on the mtDNA, the effect only operates in one direction:
It is known that nuclear gene products can and do leave the
nucleus and have an effect on mitochondrial DNA. However, the reverse is not
true – there is no evidence of mitochondrial gene products leaving the
mitochondria and having an effect on nuclear DNA.
The relationship between the nucleus and the mitochondria is
therefore one-sided. This makes it highly unlikely that donated mitochondria
could relate to the nucleus in a dysfunctional way.[72]
3.84
This view was supported by Professor Carolyn Sue, Director of the Mitochondrial
Research Centre at the Kolling Institute of Medical Research, who pointed to
other clinical situations where no haplogroup matching exists and continues without
consequence:
My feelings are that with any tissue donation—such as liver
transplants, heart transplants and bone marrow transplants—there is no
haplogroup matching, so these are models that existed in patients that I see in
hospital every day. Patients get better from their transplants. They go around
with different parts of DNA—both nuclear and, in this case, mitochondrial DNA.
But everybody forgets about that. We know that there are patients who have
mixed components of mitochondrial DNA accepting therapies, benefiting from
therapies and having their lives improved by these techniques. I see
mitochondrial donation as something like this.[73]
3.85
This view has been supported by the experimental data from the Wellcome
Centre for Mitochondrial Research in the UK that found no difference in gene
expression between the control group and the pronuclear transfer embryos.[74]
3.86
As noted above, the scientific reviews conducted in the UK recommended
that haplogroup matching be used as a precautionary step. Submitters to the
inquiry generally agreed that, in the interests of caution, mtDNA matching should
be considered.[75]
3.87
However, Professor Thorburn explained that the regulator in the UK did
not mandate that haplogroup matching must be undertaken and instead left the
decision to the families involved:
What they concluded—and I agree with it—was this should be
mentioned in discussions with the families, that there may be advantages in
matching the haplogroup but that it shouldn't be a barrier to families choosing
an unmatched donor, because it greatly restricts the number of donors that
would be potentially available.[76]
3.88
Leaving the decision to about whether to use haplogroup matching, after
counselling, to the prospective parents was endorsed by the Australian Academy
of Sciences, Murdoch Children's Research Institute and Victorian Genetic
Clinical Services and Professors Thorburn, Christodoulou, Sue, Carroll, Ryan
and Filipovska.[77]
Is an additional scientific review required?
3.89
Four safety and efficacy reviews have been conducted in the UK, the last
of which was published in 2016.[78]
The safety and efficacy reviews were carried out by an expert panel of members
who had 'no direct interests in the outcome of the review'.[79]
The scientific reviews took evidence from a range of domestic and international
experts including from the USA and the Netherlands. Based on those factors, questions
were raised at the committee's public hearing about whether there was a need
for an Australian scientific review to be conducted.
3.90
The AMDF did not consider that an Australian scientific review was
necessary:
...in terms of a suggestion of a review of the science, gauging
the public reaction and public opinion on this and looking at the ethics, that
is something that we are partly doing right now. I think the AMDF, certainly,
would draw upon the exhaustive and lengthy experience and process that was undertaken
in the UK, where three independent scientific reviews were undertaken in
relation to the science of mitochondrial donation... From the foundation's point
of view, from a science perspective, we can certainly rely on the science that
has been undertaken around the world. I don't know that there is any Australian
nuance to the science of mitochondrial disease; I don't think anything changes
around the science because we're here in Australia [80]
3.91
The Murdoch Children's Research Institute and Victorian Clinical Genetic
Services noted that Australia could adopt most of the outcomes from the process
undertaken in the UK rather than attempting to recreate the process from
scratch.[81]
Australian clinical capacity
3.92
Discussions of the safety of the science, must also take into
consideration the safety of implementing the science in the Australian clinical
context.
3.93
A number of witnesses and submitters to the inquiry indicated that the
Australian Assisted Reproductive Technology (ART) industry currently has
relevant clinical skills necessary to deliver mitochondrial donation.[82]
3.94
The Fertility Society submitted that 'Australia has been on the leading
edge of development in ART for the last 4 decades' and that in Australia 'we
are fortunate in having embryologists who have the skills and expertise to
undertake the necessary techniques to allow Mitochondrial transfer'.[83]
3.95
Professor Christodoulou submitted that the UK facility licenced to
undertake mitochondrial donation has offered to work with Australian centres
which may wish to offer these techniques, to provide any necessary instruction
or guidance.[84]
3.96
In addition to the expertise of embryologists who would undertake the
donation techniques, another ART function is the necessary counselling
procedures to ensure that potential users of this technology understand the
risks and ethics, and are therefore able to provide informed consent. The
Fertility Society submitted that the Australian ART industry 'has demonstrated
rigorous counselling and consenting processes for standard IVF treatment as
well as the more complex issues around PGD [pre-implantation genetic
diagnosis]'.[85]
Committee view
3.97
The committee understands that there are a number of possible
mitochondrial donation techniques that may be used to ensure that women living
with a mitochondrial disease do not pass it on to their children.
3.98
The committee acknowledges that these techniques – like any new reproductive
technique – involves uncertainty and a degree of risk. However, the committee
heard evidence that suggests the risks are manageable and proportionate
relative to the serious risks posed to the wellbeing of a child if it inherits
mitochondrial disease.
3.99
The committee considers that the scientific studies that have been
conducted in the UK indicate that mitochondrial donation is a procedure that
can be safely performed, and that these studies included contribution from
international experts in this field.
3.100
However, it is not the role of a Senate committee to make definitive
scientific findings. The committee therefore believes that formal endorsement
of the UK scientific findings should be made by a panel of Australian experts
with relevant scientific knowledge. This panel would be appropriately
constituted and overseen by the NHMRC.
3.101
Evidence provided to this committee indicates there are some areas for continued
scientific consideration of emerging issues such as mtDNA carrying over during
mitochondrial donation and haplogroup matching.
3.102
Whilst the committee recognises that there is still some dispute about
the potential effects of mtDNA carryover, the committee considers that it is
reasonable to consider the introduction of mitochondrial donation in Australia,
subject to further consultation.
3.103
The committee agrees that haplogroup matching requires further
scientific assessment, noting that such a step could reduce the pool of
available donors.
3.104
The committee understands that the Australian ART industry performs at
world-leading standards, and has the capacity to adapt existing skills to undertake
these techniques for safe treatment in a clinical setting. The committee also
understands that such clinics have indicated strong support for mitochondrial
donation, and are ready to support the implementation should it be made lawful.
It should be noted that these clinics could receive a financial benefit from
any legislative change that would permit mitochondrial donation in Australia.
The committee heard in evidence that local clinics could serve as a 'southern
hemisphere' hub for mitochondrial donation if this technique was legalised:
So if Australia were to follow, there would probably be two
choices—a north and a south. It would probably attract people.[86]
3.105
The committee is aware that a preferred method for mitochondrial
donation does not appear to have yet been identified. Because additional
research is being performed in this field around the world, the committee
considers that serious consideration needs to be given to the way any possible
regulation is framed to permit the safest and most up-to-date scientific
techniques to be used in Australia. The committee's views on regulatory issues
are considered in greater detail in chapter five.
3.106
The committee acknowledges that, separate to the scientific safety of
mitochondrial donation, there are significant ethical issues to consider prior
to any decision on whether to allow mitochondrial donation in Australia. These will
be discussed in the following chapter.
Navigation: Previous Page | Contents | Next Page