Chapter 3
Diagnostic testing for Lyme-like illness
3.1
As noted in Chapter 1, patients diagnosed with Lyme-like illness often
have their clinical diagnoses confirmed by laboratory tests conducted in
overseas laboratories or non-accredited laboratories in Australia. These
conflicting diagnoses cause concern and frustration to sufferers of chronic
debilitating symptoms.
3.2
This chapter examines the diagnostic process by which patients come to
be diagnosed with Lyme-like illness. It explores the discordant results for
Lyme disease testing between accredited laboratories in Australia and
laboratories overseas and non-accredited laboratories in Australia.
Diagnostic testing for Lyme disease
3.3
In 2015, the Department of Health (department) released Australian
guidelines on the diagnosis of overseas acquired Lyme disease. The department
emphasised that these guidelines are for the diagnosis of classical Lyme
disease only, and do not apply to Lyme-like illness acquired in Australia.[1]
3.4
The diagnostic protocols in the department's guidelines are consistent
with the 2014 position statement prepared by the Royal College of Pathologists of
Australasia (RCPA), Diagnostic Laboratory testing for Borreliosis ('Lyme
Disease' or similar syndromes) in Australia and New Zealand.[2]
Submissions to the inquiry from medical authorities and state and territory
governments supported the RCPA's position statement and highlighted that the
diagnostic protocol it outlines should be followed for diagnosing Lyme disease
or any similar syndromes.[3]
3.5
Figure 3.1 outlines the proposed protocol for diagnosing cases of
suspected Lyme disease in Australia recommended by the RCPA's 2014 position
statement on the treatment of Lyme disease and related syndromes.
Figure 3.1 – Recommended protocol for laboratory testing of patients with
suspected Lyme disease in Australia
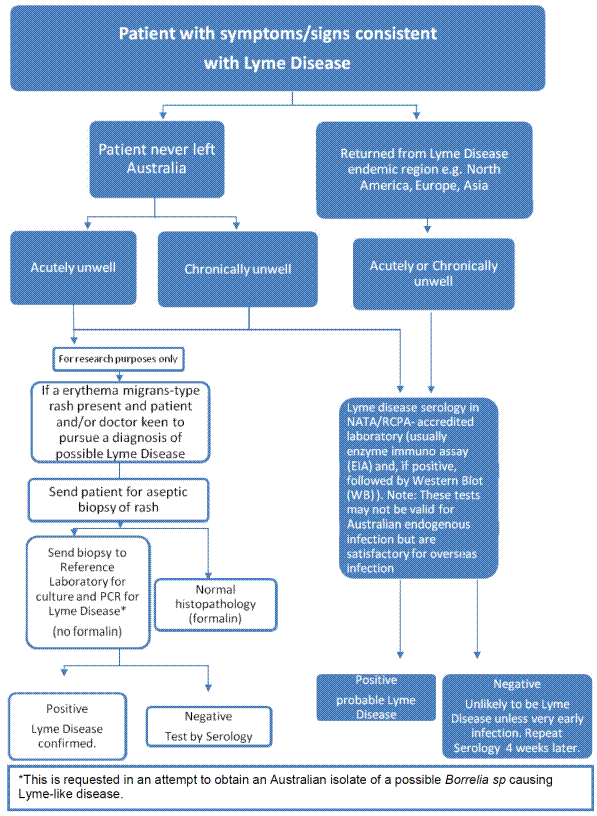
Source: Royal College of Pathologists of Australasia, Submission
532, p. 8.
Diagnosis of overseas acquired
classical Lyme disease
3.6
According to the guidelines for the diagnosis of overseas acquired Lyme
disease prepared by the department, a confirmed case of Lyme disease requires
laboratory definitive evidence (culture, DNA or serological assays), clinical
evidence and epidemiological evidence. The guidelines highlight the importance
of epidemiological evidence in determining whether a patient has Lyme disease:
Epidemiological context is important. Determining a travel
history and tick exposure prone activities are essential. The likelihood of
Lyme disease increases as the probability of a tick bite increases in a
geographically endemic area (particularly wooded, brushy, or grassy habitats).[4]
3.7
Laboratory definitive evidence for Lyme disease can be collected through
culture, DNA or serological assays. The 'gold standard' for specificity of Borrelia
infection is culture of spirochaetes from patient specimens. Molecular
detection of Borrelia bacteria using a Polymerase Chain Reaction (PCR)
test in patient specimens may also be used. However, these tests are not regarded
as reliable as the bacteria are difficult to detect and appropriate samples are
difficult to obtain.[5]
3.8
The more common way for diagnosing Lyme disease is through testing for
antibodies to Borrelia bacteria through serological assays. The United States
(US) Centers for Disease Control and Prevention (CDC) notes that serological
test results need to be interpreted according to strict criteria, including
whether Lyme disease is endemic to a particular area and whether the patient
exhibits clinical symptoms.[6]
3.9
Most serological diagnostic protocols in the US and Europe use a two
tier system. The first stage is most commonly an enzyme-linked immunosorbent
assay (ELISA), followed by a Western blot. Western blots are interpreted using
standardised criteria. These criteria differ between the US and Europe
depending on the different genospecies of B. burgdorferi in different
regions. The RCPA's position statement recommends the use of the two-tiered
system and highlights that Western blot tests 'must be interpreted with
caution, especially in the absence of an Australian Borrelia sp'.[7]
Figure 3.2 outlines the two-tiered testing process recommended by the US CDC.
Figure 3.2 – Two-tiered
testing for Lyme disease

Source: US CDC, http://www.cdc.gov/lyme/healthcare/clinician_twotier.html
(accessed 8 December 2015).
3.10
Australian laboratories are accredited for medical testing by the
National Association of Testing Authorities Australia (NATA) in conjunction
with the RCPA.[8]According
to the department, NATA accredited laboratories can readily test for Lyme
disease acquired overseas where patients have travelled to an endemic area. Dr
Gary Lum told the Community Affairs Legislation Committee:
If a patient who is from Maine, Connecticut or another area
in the north-east of the United States or from the Black Forest of Germany, who
has been bitten by a tick and then travels to visit Australia and sees a
general practitioner and has a blood test, we get a positive diagnosis. The
same is true for Australians travelling to those areas and coming back with a
rash and feeling unwell. Lyme disease is considered because they were in an
endemic area, and a diagnosis is readily made in an Australian accredited
medical testing laboratory.[9]
3.11
The Public Health Laboratory Network (PHLN) submitted that the
interpretation of serology tests depend on three key factors:
-
the sensitivity of the test (the percentage of people with the
disease who will have a positive test);
-
the specificity of the test (the percentage of people without the
disease who will have a negative test); and
-
the pre-test likelihood of the person having the disease, based
on the prevalence of the disease in the population being tested.[10]
3.12
As classical Lyme disease is considered to have a low prevalence
in Australia, locally acquired cases are considered likely to return negative
results for Borrelia. The PHLN notes that positive results for locally
acquired Lyme disease are likely to be 'false positives' and are not uncommon
in patients suffering other conditions:
... a positive result is more likely to be a false-positive if
the test is performed on a person with a low pre-test likelihood of having the
condition, such as testing for Lyme disease in Australia. There are two factors
at play here – the first is that when less stringent interpretative criteria
are used ... the results will be skewed to more patients with the disease. The
other factor is that the assays were developed for classical Lyme disease, so
for patients in a low prevalence population with nonspecific symptoms, the
predictive value is low and reactive results are more likely to reflect absence
of disease while nonreactive results likely reflect true absence of disease.
False positive results for Lyme disease are not uncommon in patients suffering
from other conditions.[11]
Diagnostic testing for Lyme-like
illness
3.13
The diagnostic protocol for testing for classical Lyme disease acquired
overseas outlined above is widely accepted by Australian medical authorities. However,
due to the debate about the cause or causes of Lyme-like illness, the
diagnostic protocol for Lyme-like illness is more disputed.
3.14
The RCPA position paper states that for patients presenting with 'syndromes
resembling Lyme disease' with no history of travel to an endemic area:
... although [i]t is not entirely possible to rule in or rule
out locally acquired Borreliosis on the basis of a series of negative results,
it is important that patients are not diagnosed erroneously as having Lyme
Disease, when they may well have some other, potentially treatable, conditions:
examples include chronic pain syndromes including fibromyalgia; complex
neurodegenerative disorders such as motor neurone disease; or psychiatric
illness such as major depression with somatisation.[12]
3.15
As noted in Chapter 1, 'Lyme literate' practitioners assert that
Lyme-like illness is caused by an ongoing Borrelia bacterial infection,
together with other co‑infections. Most other medical authorities assert
that the Borrelia responsible for causing Lyme disease is not endemic to
Australia and suggest that there may be multiple causes of Lyme-like illness
that are yet to be identified.
3.16
Many submitters who have acquired their illness in Australia stated that
when their blood samples have been sent to an accredited Australian laboratory
to test for Borrelia bacteria, the results have come back negative.[13]
3.17
However, when these same submitters consulted a 'Lyme literate'
practitioner, they have recommended sending their blood samples to either a
non-accredited laboratory in Australia or laboratories in the US or Germany. As
these tests are not covered under the Australian Medicare system, the costs for
patients are significant (for example, $800 for tests in Australia and $2 000
for tests from overseas laboratories).[14]
3.18
Tests results from these laboratories have returned a positive result
for Borrelia, often with a number of co-infections such as Bartonella
and Babesia. These results have been used by 'Lyme literate'
practitioners to confirm their clinical diagnosis. Dr Hugh Derham, a 'Lyme
literate' practitioner in Western Australia, told the committee:
Almost all of my patients have a clinical diagnosis of Lyme
disease and reasonable to excellent laboratory evidence as well, and at least
half of them have some laboratory evidence from an accredited laboratory,
either accredited by or recognised by NATA. I do not have hundreds of patients
who believe they have Lyme disease; their belief is founded on good evidence.[15]
3.19
Evidence from submissions suggests that the differences between
laboratory results can cause significant confusion and frustration for
patients. Submitters expressed their deep concern that results from overseas
laboratories are disregarded by Australian medical authorities, particularly
infectious disease specialists:
Patients who attempt IDS [infectious disease specialist]
consults are turned away with negative test results even though they may have
gone to huge expense to be tested in accredited laboratories overseas and carry
positive test results with them, they are still disregarded by the IDS.[16]
3.20
The issue of discordant results between accredited laboratories in
Australia and non-accredited Australian and overseas laboratories needs further
inquiry.
Accreditation of Australian
laboratories
3.21
The committee heard that one non-accredited Australian laboratory,
Australian Biologics, is used by a number of 'Lyme literate' practitioners to
test for Lyme-like illness. In her evidence to the committee, the Director of
Australian Biologics, Ms Jennie Burke stated that through their testing
process, Australian Biologics has identified evidence of Borrelia in
Australian paralysis ticks.[17]
3.22
Unlike other Australian accredited laboratories, Australian Biologics
uses Polymerase Chain Reaction (PCR) assays to test for the presence of Borrelia
DNA in human samples. Australian Biologics also uses different serological
tests for Borrelia imported from Germany (the Mikrogen recomLine
and AID EliSpot Lymphycyte Transformation Test). In their submission,
Australian Biologics asserts that the serological tests used by other
Australian laboratories are not effective for patients with a chronic infection
of Borrelia and that PCR and the German serological tests are more
effective.[18]
A large number of submitters who have been diagnosed with Lyme-like illness
have noted that they have had positive tests for Borrelia from
Australian Biologics.[19]
3.23
A number of submissions have expressed concerns about the reliability of
the tests provided by Australian Biologics. For example, one infectious disease
specialist suggested that the false positive rate for the tests used by
laboratories such as Australian Biologics 'appears to be extremely high'.[20]
3.24
Australian medical authorities noted that results from laboratories that
are not accredited by NATA and the RCPA, such as Australian Biologics, should
be interpreted with caution. The RCPA submitted:
If a laboratory is not NATA/RCPA accredited this means that
the laboratory may not have testing protocols and quality assurance processes
in place that would be considered satisfactory compared to the standards. Such
laboratories may be more likely to obtain an incorrect result for a particular
laboratory investigation.[21]
3.25
However, Ms Burke expressed concerns about the process by which
laboratories are accredited by NATA. Australian Biologics applied for
accreditation in 2014 for its PCR testing. Ms Burke stated that NATA does not
acknowledge the accuracy of their testing protocols for Borrelia or
their quality assurance programs, and that it breached its charter in assessing
their accreditation application by disclosing confidential intellectual
property information to a rival laboratory.[22]
3.26
In evidence to the committee, representatives from NATA highlighted that
in the accreditation process for innovative laboratory processes, such as PCR
testing for Borrelia, the threshold for evidence is higher than for
usual accreditation:
For new and innovative methods for which the availability of
appropriate validation is limited or where standard methods have been modified
or, indeed, used outside their design parameters, the threshold of evidence for
acceptance naturally becomes higher. The soundness of evidence provided is
judged by relevant experts and professional bodies, not by employees of NATA.
NATA must seek the best advice from expert sources, peers of the laboratory,
before it commits to a precedent that will impact on the health and safety of
the Australian population.[23]
Recognition of overseas laboratories
3.27
The committee heard arguments from 'Lyme literate' practitioners that
the tests for Borrelia conducted by accredited Australian laboratories
are not appropriate, and the criteria by which they are interpreted are
inadequate. These practitioners assert that the two-tier process recommended by
the RCPA and the US CDC does not adequately detect Borrelia and other
co-infections acquired in Australia.[24]
3.28
For example, Dr Peter Dobie from the Australian Chronic Infectious and
Inflammatory Disease Society told the committee that the ELISA test – the first
tier in the two tier process – is not sensitive enough to detect Lyme-like
illness and should be 'abandoned':
...most Australian pathology laboratories are doing the wrong
blood test for Lyme disease. This is one reason why Lyme disease and Lyme-like
illness are underdiagnosed in Australia. Most laboratories are using a test
called the ELISA test. This test is not sensitive enough to detect most cases
of this illness ... Pathology laboratories should be doing western blot and PCR
as the frontline tests for Lyme disease, not the ELISA test.[25]
3.29
These practitioners insist that overseas laboratories (specifically
IGeneX in California and Arminlabs or Infectolab in Germany) are better placed
to accurately test for Borrelia. Dr Dobie told the committee that the
main reason that 'Lyme-literate' practitioners use overseas laboratories is
that these laboratories will do the Western blot test if requested, whereas
Australian laboratories will only do so if the ELISA test is positive:
One of the reasons that doctors are using these overseas
laboratories is that these laboratories will do the western blot on request.
Doctors treating this illness are not interested in the result of the ELISA
test. If Australian laboratories would do the western blot on request, there
would be less need for us to use overseas laboratories.[26]
3.30
The committee also heard that the US CDC criteria used to interpret
serological tests in accredited Australian laboratories focus too narrowly on Borrelia
burgdorferi. Dr Mualla McManus told the committee that the CDC criteria are
not appropriate for identifying other possible Australian species of Borrelia:
The government only thinks of Lyme disease, and follows the
CDC criteria ... We have Borrelia burgdorferi, and a subset of that is
Lyme disease. We have relapsing fever, and it has over 20 genospecies already.
We have reptilian borrelia, but the infection has not yet been found in humans.
So if we concentrate on Lyme disease we are missing out on 80 per cent of other
borrelia infections, and that is really dangerous. We are being short-sighted ...
We could have a unique class of borrelia.[27]
3.31
Some submitters suggested that results from overseas laboratories should
be interpreted with caution, as each test has its own sensitivity and
specificity based on the composition of the causative agent. According to these
submitters, in the absence of a known causative agent in Australia, a positive
test result is likely to indicate a false positive due to cross reactions from
other bacteria.[28]
The RCPA submitted:
If caused by a tick-born microbe, the causative microbe has
not yet been identified and thus its antigenic make-up is unknown. Without knowing
its antigenic make-up, it is impossible to design a proper serological test
with measurable sensitivity and specificity. Cross-reactivity between patient
antibodies and Borrelia antigens from overseas Borrelia used in vitro in
Australian diagnostic assays are hard to predict.
There are many species of spirochetes (including Borrelia
spp.) present in the normal human gastrointestinal tract (including the
oral cavity) and some of these may potentially cause cross-reacting antibodies
to be produced by the patient.[29]
3.32
Some submitters also suggested that tests conducted in non-NATA
accredited laboratories in Australia and laboratories overseas may produce
different results to accredited Australian laboratories because they may not
interpret their results according to the criteria set by the US CDC and the
European Society of Clinical Microbiology and Infectious Diseases.[30]
Dr Lum told the committee:
... when these tests are performed overseas, and also in some
specialist laboratories in Australia, the interpretive criteria are different.
What I mean by that is that they place less serological stringency on the test
interpretation, so it makes it easier to diagnose a reactive [positive] result.[31]
3.33
The RCPA cautioned that it is difficult to assess the accuracy of
results from serological tests conducted in overseas laboratories that are not
accredited to Australian standards:
Overseas laboratories are by definition, not accredited to
Australian standards so their use by Australian doctors and patients is on the
basis of unknown quality of testing. While some may be excellent laboratories,
accredited to international and their own country's standards and producing
accurate and precise results, others may not be so. Australian authorities are
not in a position to regulate or monitor these overseas laboratories and it is
very difficult for Australian clinicians and patients sending specimens
overseas to assess what veracity to place on the results and reports that they
receive.[32]
3.34
The RCPA also warned that the overseas laboratories favoured by 'Lyme
literate' practitioners are not used by 'mainstream' practitioners in their own
countries and are likely to return false positive results. Professor Stephen
Graves from the RCPA told the committee:
The laboratories in Germany and the United States that you
are talking about, and that we are talking about now, are a minority. They are
an exception. The mainstream doctors in those countries do not use those
laboratories. They do not use them because they give them the wrong result.
They give them false positive results. So it is not just us. The doctors in
those countries say, 'Don't send your stuff to such and such a laboratory; you
can't trust the result.' People here who are not getting results from
mainstream laboratories are sending them to very off-the-mainstream types of
laboratories in other countries. They are not the mainstream laboratories that
are doing the routine testing all the time.[33]
3.35
'Lyme literate' practitioners suggested that NATA, the body responsible
for accrediting Australian laboratories, should recognise the overseas
accreditation of these specific laboratories overseas, through such measures as
the International Laboratory Accreditation Cooperation Mutual Recognition
Arrangement (MRA).[34]
Some advocacy groups also suggested that NATA should acknowledge that the
overseas laboratories in question are accredited to the international standards
for medical testing (ISO 15189) and should therefore recognise results from
these laboratories. For example, Ms Rebecca Vary from the Lyme Disease
Association of Australia (LDAA) suggested to the committee that NATA should
recognise results from a German laboratory, Infectolab:
Infectolab were accredited with ISO 15189, and they have been
accredited to that standard for quite a while. What happened in January was
that NATA became a member of that accreditation as well, so NATA now has the
right to recognise the other labs in ISO 15189, so it can therefore recognise
the Infectolab results.[35]
3.36
In evidence to the committee, representatives from NATA confirmed it had
achieved international recognition for medical testing (ISO 15189) in January
2016 under the MRA. However, NATA emphasised that the effect of MRA recognition
is of equivalence of overseas testing methods; it does not expect or require
laboratories or medical authorities in Australia to recognise another country's
specific requirements or context:
Our role under the MRA is to promote recognition of
equivalence. It is the end user, however, who is actually the individual making
the final decision on the recognition.[36]
Limits on diagnostic testing under
Medicare
3.37
The committee also heard that under the current Medicare Benefits
Schedule, laboratories are only able to test for what the referring doctor
requests. The Australian Rickettsial Reference Laboratory suggested that these
limits mean that when a patient sample is referred for a Lyme disease test,
laboratories are not be able to test for other known pathogens:
Diagnostic tests on patient specimens are generally
bulk-billed and the income from this [85% of the Medical Benefit Schedule
recommended fee] only covers the cost of undertaking the test requested by the
referring doctor. Thus if the doctor asks for investigations for Lyme Disease,
the laboratory is unable to also test for other potentially tick-transmitted
diseases, despite the possibility that the patient may have acquired one of
these, eg a rickettsial infection, which the referring doctor did not include
on his/her list of differential diagnoses.[37]
3.38
Professor Stephen Graves from the RCPA and also Director of the Australian
Rickettsial Reference Laboratory, suggested that the Medicare rules around
laboratory testing should be changed to allow laboratories to explore other
possible diagnoses when a test is referred for Lyme disease:
...what I am proposing ... is somehow make it possible for
diagnostic laboratories—the sort of laboratory that is part of the Public Health
Laboratory Network, like my laboratory, the Australian Rickettsial Reference
Laboratory. If we get a serum specimen in from a patient who has query Lyme
disease—endemic Australian Lyme disease—we can currently only test for Lyme
disease. That is all I am allowed to do. If I do any other testing, it is
basically called overservicing and, as a pathologist, I can get into big
trouble over it. So I just have to do what is requested. So I do the Lyme
disease testing. It is negative—end of story. But if I could also test for
Coxiella, Rickettsia, Anaplasma, Ehrlichia, Neoehrlichia—although we do not
have an assay for that yet—Bartonella or Babesia, that would make a big
difference. We could possibly find out what is affecting these people. But not
only cannot we do it; we are not allowed to do it.[38]
3.39
The department confirmed that there are 'coning' rules in place that
only allow laboratories to seek remuneration for up to three tests under the
current Medicare rules:
...under the current way that pathology testing is remunerated,
that there not be any sense of overservicing, but if there is a legitimate
request then there will not be overservicing. The important thing to remember,
though, is that the pathology profession is subject to various rules under the
Medicare Benefits Schedule and, when referrals are made by general
practitioners, there are rules in place which make it difficult compared to
when, say, another specialist medical practitioner makes a referral, such that
the ability to make a claim on those tests is different. That needs to be
understood. For example, if a general practitioner requests more than three
tests, there is a coning rule in place, and the pathology practice will only
receive remuneration for the most expensive three tests, rather than all of the
tests.[39]
Measures to address discordant
results
3.40
To address the discordant results between overseas laboratories and
accredited laboratories in Australia, Professor John Mackenzie, author of the
2013 scoping study, suggested that the department coordinate a quality
assurance/quality control (QA/QC) assessment of overseas testing procedures:
... the use of an international panel of specimens should be
used for QA/QC is an essential step, and any putative positives should then be
investigated fully in collaboration with the laboratory which has made the
positive claim, and in parallel and together with an accredited public health
reference laboratory to substantiate the claim ... The DOH [Department of Health]
should indeed urgently liaise with overseas laboratories which claim to find
positive results to ensure they participate in a QA/QC assessment, and to
ensure this is carried out properly, an international accredited and unaligned
laboratory such as the UK Institute for Biological Standards and Control should
be engaged to oversee the conduct and interpretation of the QA/QC results.[40]
3.41
The committee heard that the department is currently investigating the
different approaches to the laboratory diagnosis of Lyme disease worldwide. The
department has contracted the National Serology Reference Laboratory[41]
to:
... evaluate the serological assays used to diagnose Lyme
disease in specialist laboratories in Australia and overseas as well as
accredited pathology laboratories in Australia. The specimens being tested are
from individuals in Australia and overseas both with and without symptoms. The
results will be used to examine the performance characteristics of these
laboratory tests and hopefully resolve the conundrum of discordant results in
laboratories in Australia and overseas.[42]
3.42
The committee notes that the department has previously advised the
Senate Community Affairs Legislation Committee that ten laboratories have been
approached and invited to participate in this evaluation, which is due to
report in early 2017.[43]
3.43
The department suggested that it would welcome a review of current
laboratory testing processes and treatments by the Medicare Services Advisory
Committee and the Pharmaceutical Benefits Advisory Committee. The department
submitted:
Both committees are in the best position to review the
current data for the available diagnosis and treatment. Should the committees
advise that supportive evidence of effectiveness and cost-effectiveness does
exist, steps can be taken to update the Medicare Benefits Schedule and the
Pharmaceutical Benefits Schedule.[44]
Committee
view
3.44
The committee is aware that discordant laboratory results between
accredited laboratories in Australia and non-accredited Australian and overseas
laboratories cause confusion and frustration for patients.
3.45
The committee supports the department's work with the National Serology
Reference Laboratory to conduct an evidence-based assessment of laboratory
testing in Australia and overseas, with a focus on tests for Lyme-like illness.
3.46
The committee notes that the following issues need further inquiry:
-
progress of the National Serology Reference Laboratory's
assessment of Australian and overseas laboratory testing for Lyme-like illness;
-
the process of laboratory recognition and accreditation to assist
patients in understanding why there are discordant results; and
-
options for changing the Medicare rules to allow accredited
Australian laboratories to explore possible alternate pathogens.
Navigation: Previous Page | Contents | Next Page